Physics Modern Physics Level: High School
A hypothetical atom has four energy states as shown in the figure at “click here" Which of the following photon energies Ev could not be found in the emission spectra of this atom after it has been excited to the n = 4 state ?
1) Ev = 5 eV
2) Ev = 4 eV
3) Ev = 1 eV
4) Ev = 3 eV
5) Ev = 2 eV

Physics Modern Physics Level: High School
Dual Nature
An electron and a photon each have a wavelength of 0.1 nm . Compare their kinetic energies .

Physics Modern Physics Level: High School
Dual Nature
An athlete weighing 70 kg travel at a speed of 10 m/s . What is her de Broglie wavelength ?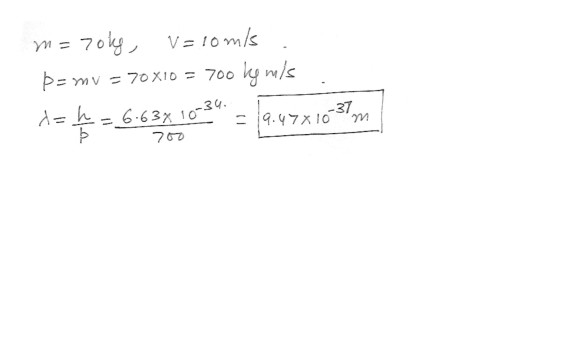
Physics Modern Physics Level: High School
Dual Nature
Gold has a work function W of 4.9 eV .a) What is the minimum frequency of photon needed to observe the photoelectric effect ?
b) If illuminated by ultraviolet radiation of frequency 2.3 x 10^15 Hz what is the K.E in Joules of the emitted electron ?
c) What is the energy in electron volts ?

Physics Modern Physics Level: High School
Dual Nature
An electron is accelerated through a potential difference of 1000V what is .a) Its K.E in electron volts ?
b) Its K.E in Joules ?
c) Its speed ?
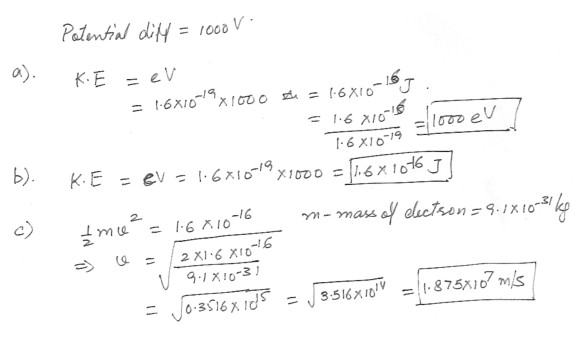
Physics Modern Physics Level: High School
Dual Nature
Calculate the energy of(a) a photon of frequency 4.5 x 10^14 Hz .
(b) a photon of wavelength 6.0 x 10^-7 m
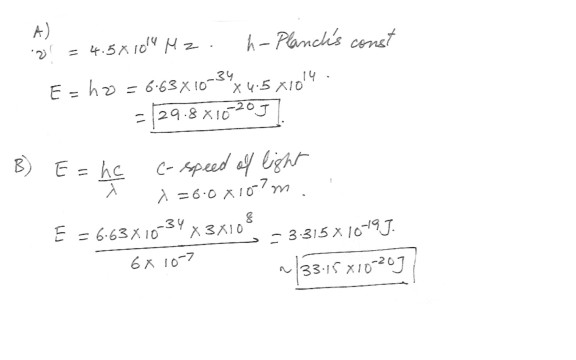
Physics Modern Physics Level: High School
Bohr’s Atomic Model
The electron of a hydrogen atom is initially at the orbit n = 4 , If this electron first goes to the orbit n = 3 and then to the orbit n = 2 and then the orbit n =1 , how many photons are emitted during these transitions ? How many photons are emitted if the electron directly falls from the orbit n = 4 to the orbit n = 1 ?
Physics Modern Physics Level: High School
Photoelectric Effect
The work function for cesium is 2.0 eV . Find the maximum speed of the ejected electrons when a cesium surface is illuminated by violet light of wavelength 4.5 x 10 ( to the -7) m .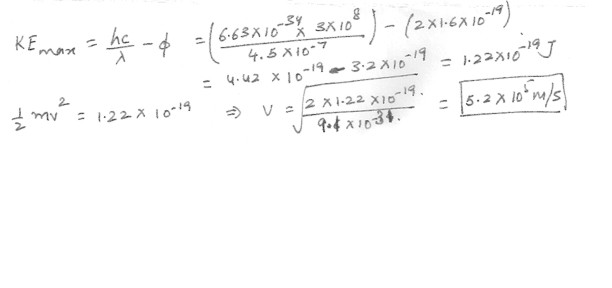
Physics Modern Physics Level: High School
Dual Nature
The human eye can detect as few as three photons of 5.6 x 10 (to the -7) m light arriving together . How much energy do these three photons collectively represent ?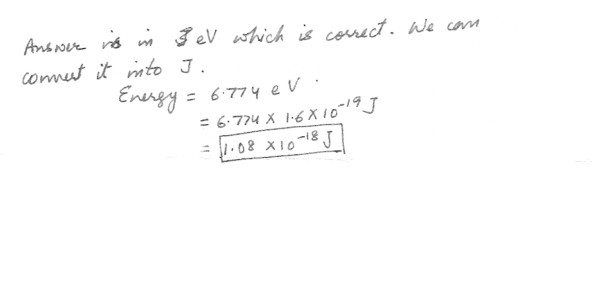
Physics Modern Physics Level: High School
Heisenberg’s Uncertainty Principle
Find the minimum uncertainty in a proton’s velocity if this proton is confined in a box of length 5 x 10 (to the -8) m .
Physics Modern Physics Level: High School
Dual Nature
How many photons are emitted each second by a 1000 Watt source that emits electromagnetic waves at the wavelength of 0.05 m ?
Physics Modern Physics Level: High School
Dual Nature
X-ray photons having a wavelength of 0.267 nm are directed at a tisse spe4cimen . For radiation which is Compton scattered at an angle of 65degrees from the original direction, calculate the followinga) The Compton wavelength shift
b) The wavelength of th scattered photons
c) The energy of the original photon
d) The energy of the scattered photon
e) The energy of the recoil electron
f) The direction of scatter to produce a maximum Compton wavelength shift
g) The maximum Compton wavelength shift

Physics Modern Physics Level: High School
Nuclei
What is the binding energy per nucleon for tritium 3H which is the heaviest isotope of hydrogen ?
Physics Modern Physics Level: High School
a) For the “P” electrons in the L shell , list the details of each possible combination of quantum numbers possible and deduce the total number of electrons possible in the “2p” subshell .
b) How many electrons can occupy the M shell ? Explain with details .

Physics Modern Physics Level: High School
Nuclei
What are the number of neutrons and protons for the following nuclides : 6Li, 13C, 40K and 64Cu .
