Physics Properties Of Matter Level: High School
Equation of Continuity
Calculate the mass flow rate (in g per second ) of blood (density = 1.0 g/cm^3) in an pipe with cross-sectional area of 2.0 cm^2 if the flow speed is .4 m/s . Assume that the aorta branches out into capillaries with a cross sectional area of 3 x 10^3 cm^2 , what is the flow speed in the capillaries ?
Physics Properties Of Matter Level: High School
Pressure
When the two arms of a U-tube manometer containing mercury are connected to two chambers , the difference in the heights of mercury in the two arms is 4.5 cm . What is the difference in pressure between the chambers ?a) 6.0 x 10^5 Pa
b) 1.0 x 10^4 Pa
c) 1.0 x 10^5 Pa
d) 6.0 x 10^3 Pa
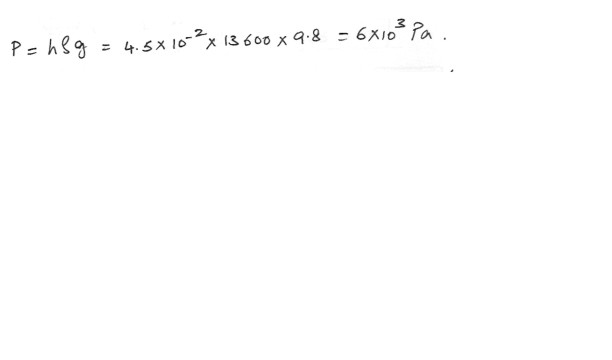
Physics Properties Of Matter Level: High School
Archimedes Principle
A piece of wood floats in oil of density 800 kg/m^3 with 25% of its volume above the surface of the oil . If the piece of wood is placed in water , it will float in water witha) 75% of its volume above the water
b) 60 % of its volume above the water
c) 25% of its volume above the water
d) 40% of its volume above the water
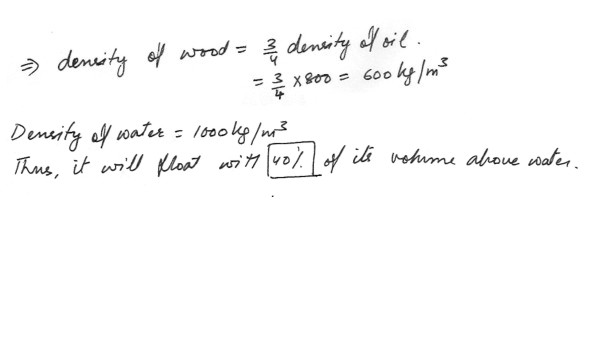
Physics Properties Of Matter Level: High School
Elasticity
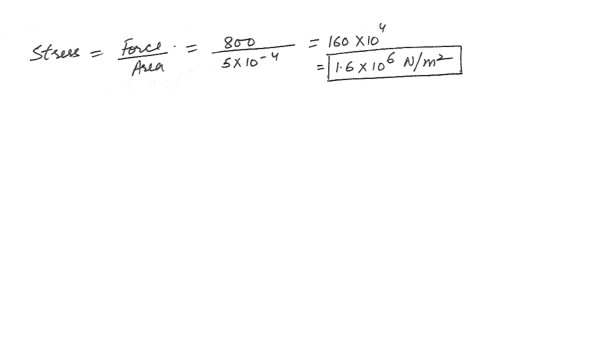
A cylindrical specimen of steel of cross-sectional area 5 cm^2 is objected to a tensile force of 800 N . The stress in the material is :a) 3.2 x 10^6 N/m^2
b) 8.0 x 10^3 N/m^2
c) 4.0 x 10^3 N/m^2
d) 1.6 x 10^6 N/m^2
Physics Properties Of Matter Level: High School
Thermal Expansion
A thin rod contains of two parts joined together . One-half of it is silver and one-half is gold . The temperature decreases by 24 degrees C . Determine the fractional decrease in the rod’s length using the formula ‘delta LI(Lo, sivlever + Lo,gold) , Where ‘Lo, silver’ and ‘Lo, gold’ are the initial lengths of the silver and gold rods .)
Physics Properties Of Matter Level: High School
Thermal Expansion
A rod made from a particular alloy is heated from 23.5 degree to the boiling point of water . Its length increases by 8.51 x 10^-4 m . The rod is then cooled from 23.5 degrees C to the freezing point of water . By how much does the rod shrink ?
Physics Properties Of Matter Level: High School
A container has a large cylindrical lower part with a long thin cylindrical neck ( see the figure at ‘click here’ ) The lower part of the container holds 12.5 m^5 of water and the surface area of the bottom of the container is 5.00 m^2 . The height of the lower part of the container is 2.50 m and the neck contains a column of water 8.50 m high . The total volume of the column of water in the neck is 0.200 m^3 . What is the magnitude of the force exerted by the water on the bottom of the container ?
Physics Properties Of Matter Level: High School
A nurse applies a force of 4.40 N to the piston of a syringe . The piston has an area of 5.00 x 10^-5 m^2 , what is the pressure increase in the fluid within the syringe ?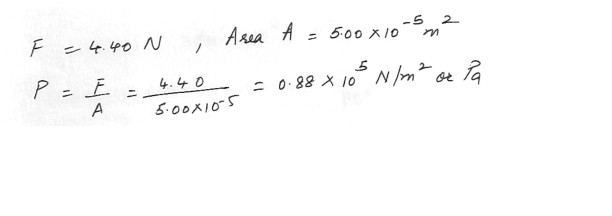
Physics Properties Of Matter Level: High School
An idea gas with internal energy U at 200- degrees C is heated to 400- degrees C . Its internal energy then will be :a) still U
b) 2 U
c) 1.4 U
d) 1.2 U
e) 4.1 U
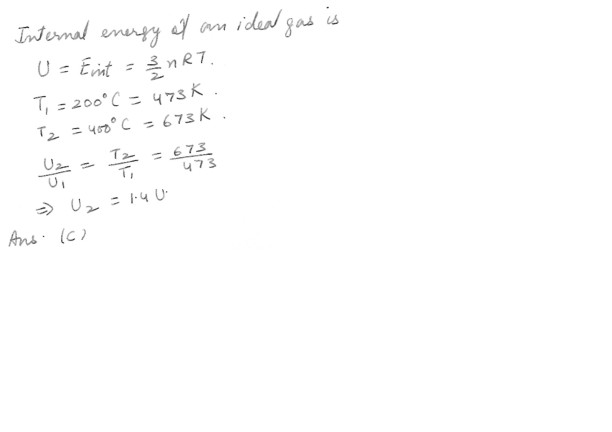
Physics Properties Of Matter Level: High School
Gases
Oxygen molecules are 16 times more massive than hydrogen molecules . At a given temperature , how do their average molecular speeds compare ? The oxygen molecules are moving .a) 4 times faster
b) at ¼ the speed
c) 16 times faster
d) at 1/16 the speed
e) none of the above
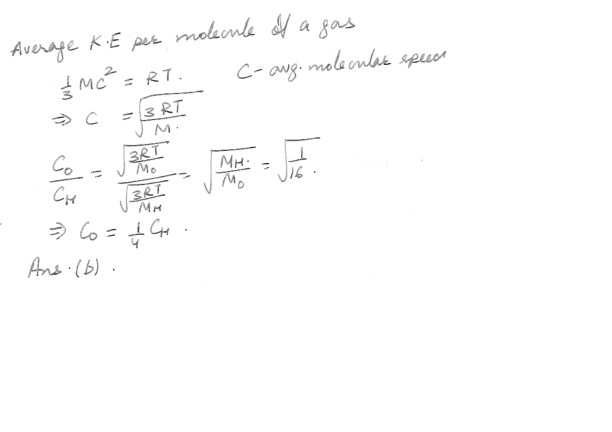
Physics Properties Of Matter Level: High School
Gases
A sample of an ideal gas is heated and its Kelvin temperature doubles . What happens to the average speed of the molecules in the sample ?a) It does not change
b) If doubles.
c) It halves.
d) It increases exponentially .
e) None of the above.
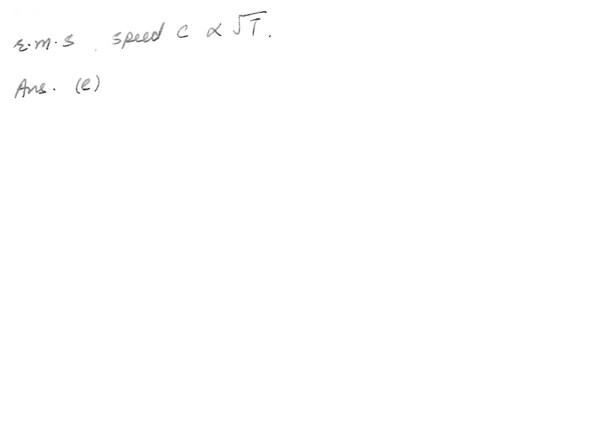
Physics Properties Of Matter Level: High School
The number of molecules in one mole of a substance :a) depends on the molecular weight of the substance .
b) depends on the atomic weight of the substance .
c) depends on the density of the substance .
d) depends on the temperature of the substance.
e) is the same for all the substance
Physics Properties Of Matter Level: High School
Gases
According to the ideal gas Law , PV = constant for a given temperature . As a result , an increase in volume corresponds to a decrease in pressure . This happens because the molecules :a) collide with each other more frequently .
b) move slower on the average .
c) strike the container wall less often .
d) transfer less energy to the walls of the container each time they strike it .
e) equal Avogadro’s number .
Physics Properties Of Matter Level: High School
Gases
A container of an ideal gas at 1 atm is compressed to one-third it volume with the temperature held constant .What is its final pressure ?a) 3 atm
b) 1 atm
c) 1/3 atm
d) 9 atm
e) 273 atm

Physics Properties Of Matter Level: High School
Thermal Expansion
Consider a flat steel plate with a hole through it center . When the plate’s temperature is increased , the hole will :a) expand only if it takes up more than half the plate’s surface area .
b) contract if it takes up less than half the plate’s surface area .
c) always contract
d) always expand
e) none of the above
