Physics Properties Of Matter Level: High School
What is the net upward force on an airplane wing of area 20 m^2 if the speed of flow is 300 m/s across the top of the wing and 280 m/s across the bottom ?
Physics Properties Of Matter Level: High School
A steel safe of weight 1.00 x 10^4 N . is being raised to the top of a building. The steel cable to be used was only 15 m long and so an additional 7 m length of another cable had to be used . If both cables have a diameter of 1.00 cm , and the total stretch of both cables put together is 2.35 cm , What is the Young’s modulus of the second cable ? The weight of both cables is negligible compared to that of the safe .
Physics Properties Of Matter Level: High School
Pressure
Suppose that a girl weighing 125 pounds is wearing “spike” heel shoes . If she surface of the heel in contact with th floor corresponds to a square measuring ¼ on each side , and if she stands momentarily with all her weight on one heel , what is the pressure she exerts on the floor ?125 lbs/in^2
500 lbs/in^2
1000 lbs/in^2
1250 lbs/in^2
2000 lbs/in^2
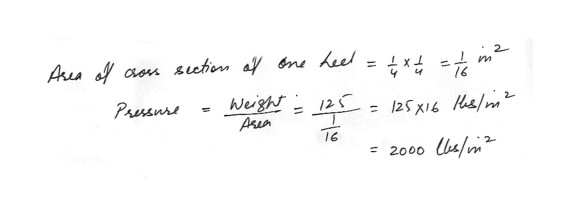
Physics Properties Of Matter Level: High School
Archimedes Principle
A glass container is half-filled with mercury , and a steel ball is floating on the mercury . If water is then poured on top of the steel ball and mercury , filling the glass , What will happen to the steel ball ?a. It will float higher in the mercury .
b. It will float at the same height . v
c. It will float lower in the mercury .
d. It will sink to the bottom of the mercury .
when water is poured , there will be an up thrust (or apparent loss in weight) of the steel ball due to the water displaced by it . Thus , steel ball will becomes slightly lighter and so it will displace less of Hg to counter balance its weight .
Physics Properties Of Matter Level: High School
Elasticity
When a force F is applied to a certain rod , there is a 1 cm change in length . If that same force is applied to a rod that is three times bigger in each dimension , what will be the change in length ?9 cm
3 cm
1 cm
0.33 cm
0.667 cm

Physics Properties Of Matter Level: High School
A large stone is resting on the bottom of the swimming pool . The normal force of the bottom of the pool on the stonea. is equal to the weight of the stone
b. is equal to the weight of the water displaced
c. is equal to the sum of the weight of the stone and the weight of the displaced water.
d. is equal to the difference between the weight of the stone and the weight of the displaced water.
Physics Properties Of Matter Level: High School
Which of the following states of matter is associated with the very highest of temperatures ?Liquid
Plasma
Gas
Solid
Physics Properties Of Matter Level: High School
Write Bernoulli’s equation for fluid flow . Give some example of types of problems that this equation may be applied to , and review the conditions under which some of the terms of the equation may cancel out or be equal to zero . Thus simplifying the equation .
Physics Properties Of Matter Level: High School
Fluids
What is meant by flow rate in fluids ?Physics Properties Of Matter Level: High School
Fluid
Are there some simplifications you can make to Bernoulli’s equation to help solve some common types of problems ? Include diagrams .

Physics Properties Of Matter Level: High School
Hydrostatic pressure
Water is filled to height H behind a dam of width w (see figure at “click here”) . Determine the resultant force exerted by the water on the dam . (Hint: The pressure is not constant for this problem , it varies with depth .)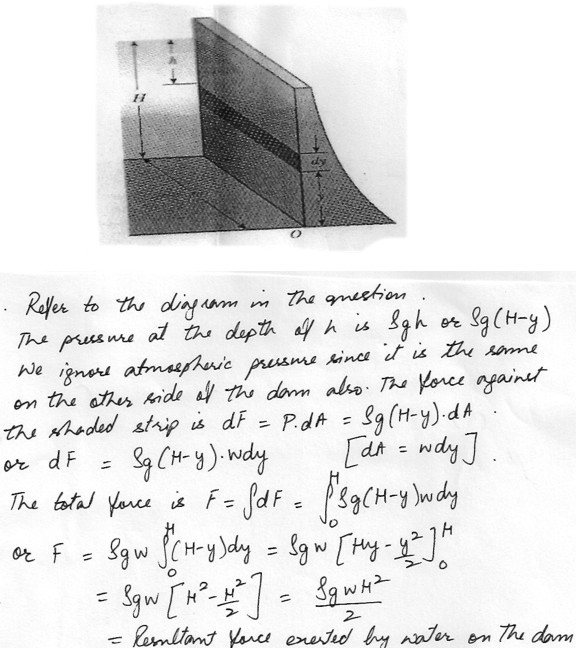
Physics Properties Of Matter Level: High School
Elasticity
The weight that will cause a wire of diameter d to stretch a given distance , for a fixed length of wire , isProportional to d^2
Independent to d
Independent of what the length of the wire is
Proportional to d
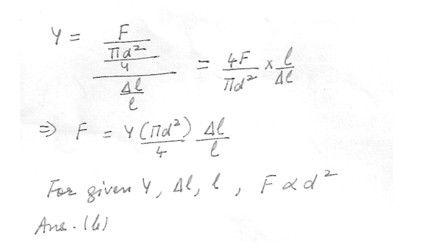
Physics Properties Of Matter Level: High School
Which two temperature changes are equivalent ?1 degree F = 1 degree C
1 degree C = 1 K
1 K = 1 degree F
Physics Properties Of Matter Level: High School
Gases
Both the pressure and volume of a given sample of an ideal gas double . This means that its temperature in Kelvin must .Quadruple
Reduce to one-fourth its original value .
Reduce to one-half its original value .
Remain unchanged
Double
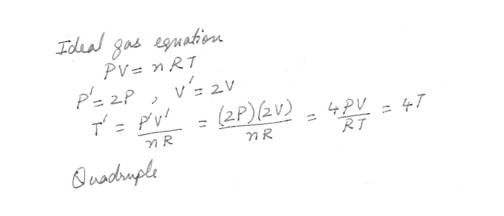
Physics Properties Of Matter Level: High School
Gases
The kinetic theory of gases suggests that the average kinetic energy per molecule of an ideal (mono-atomic)gas is .kT
3/2 kT
2 kT
1/2 kT
