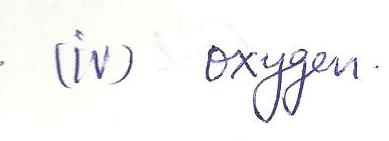Chemistry Inorganic Chemistry Level: Misc Level
The two suffixes that indicate the presence of oxygen in a polyatomic ion are
a.-ite &- ide
b. - ite & -ide
c. -ate &- ide
d.- thio &- penta
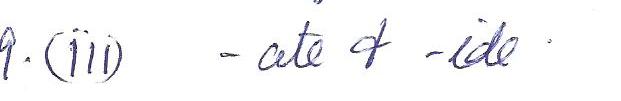
Chemistry Inorganic Chemistry Level: Misc Level
The lewis structure for methane, CH4, has
a. 2 double bonds
b. 1 triple bond
c. 4 single bonds
d. 2 single bonds & 2 double bonds

Chemistry Inorganic Chemistry Level: Misc Level
the correct formula for Magnesium Oxide is
a.MgO2
b.MgO
c Mg2O
d.Mg2O3

Chemistry Inorganic Chemistry Level: Misc Level
VSEPR theory staes that the repulsion between electron pairs surrounding an atom causes
a.these pairs to be as far apart as possible
b.an electron sea to form
c.a bond to break
d.a covalent bond to form

Chemistry Inorganic Chemistry Level: Misc Level
the charge of an ion is
a. always positive
b. always negative
c.either positive or negative
d.zero

Chemistry Inorganic Chemistry Level: Misc Level
The tendency of atoms to gain or lose electrons so that their ouer orbitals are filled with electrons is the
a.vseper theory
b. Quartet rule
c.Octet Rule
dElectronegativity rule

Chemistry Inorganic Chemistry Level: Misc Level
Compounds that are electrically neutral must have equal numbers of
a.anions &cations
b.positive & negative charges
c.moleecules
d.ionic bond
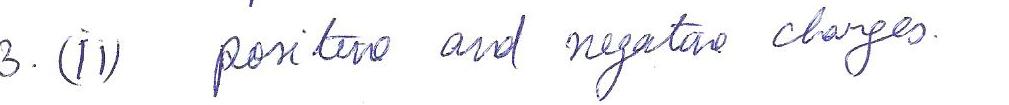
Chemistry Inorganic Chemistry Level: Misc Level
A covalent bond in which two atoms share three pairs of electrons between is called a (n)
a. single bond
b. double bond
c.triple bond
d.metallic bond

Chemistry Inorganic Chemistry Level: Misc Level
A polar covalent bond is likely to form between two atoms that
a.differ in electronegativity
b. are of simliar size
c. differ in state of matter
d. have the same number of electrons

Chemistry Inorganic Chemistry Level: Misc Level
an ion that has a similar electron configuration to that of neon is
a.CI-
b. Na
C. Na+
d.O_
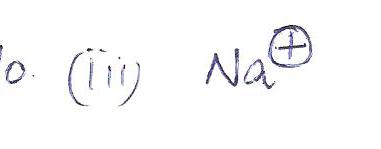
Chemistry Inorganic Chemistry Level: Misc Level
According to the octet, a calcium atom has a tendency to
a.lose one electron
b.lose two electrons
c.gain one electron
d.gain two electrons
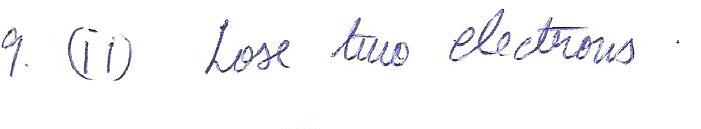
Chemistry Inorganic Chemistry Level: Misc Level
The correct Lewis structure for the boron atom has
a.one single valence electron
b.two single valence electron
c.three single valence electrons
d. one pair of electrons and 3 single valence electron
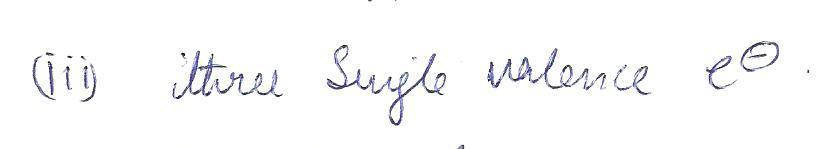
Chemistry Inorganic Chemistry Level: Misc Level
lonic bonding occurs between
a.ions of opposite electrical charge
b.ions of the same electrical charge
c. ions of the same size
d. ions of the same size and opposite charge
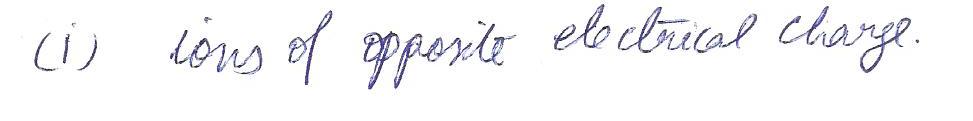
Chemistry Inorganic Chemistry Level: Misc Level
The correct name for the salt with the formula CaI2 is
a. calcium iodie
b.calcium di-iodine
c. calcium iodine
d. dicalcium iodide
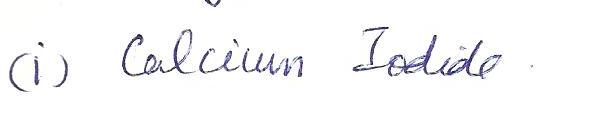
Chemistry Inorganic Chemistry Level: Misc Level
Multiple covalent bonds may be found in the atoms that contain carbon, nitrogen, or
a. helium
b. argon
c. hydrogen
d. oxygen